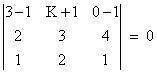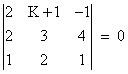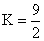Chemical EquilibriumHard
Question
If the lines 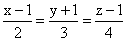 and
and 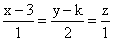 intersect, then k is equal to:
intersect, then k is equal to:
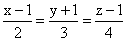 and
and 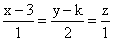 intersect, then k is equal to:
intersect, then k is equal to:Options
A.0
B.- 1
C.2/9
D.9/2
More Chemical Equilibrium Questions
At a temperature T, a compound AB4(g) dissociates as 2AB4(g) ⇋ A2(g) + 4B2(g) with a degree of dissociation x, whi...For the reaction 2NO2(g) ⇋ 2NO(g) + O2(g), (Kc = 1.8 × 10-6 at 184oC) (R = 0.0831 kJ / (mol.K) When KP and Kc...For the reaction SO2(g) + O2(g) ⇋ SO3(g), if KP = KC(RT)x where the symbols have usual meaning then the value of x...The value of Kp for the reaction, 2H2O(g) + 2Cl2(g) ⇋ 4HCl (g) + O2 (g)is 0.03 atm at 427oC, when the partial pres...What can you say about the existance of A if the potential energy diagram for the reaction A → B looks like...