Chemical EquilibriumHard
Question
At a temperature T, a compound AB4(g) dissociates as 2AB4(g) ⇋ A2(g) + 4B2(g) with a degree of dissociation x, which is small compared with unity. The expression of KP in terms of x and total pressure P is ;
Options
A.8P3x5
B.256P3x5
C.4Px2
D.None of these
Solution
2AB4(g) ⇋ A2(g) + 4B2(g) ∑ n = 1 + 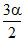 ≃ 1
≃ 1
1 - α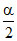 2x 1 - α ≃ 1
2x 1 - α ≃ 1
KP =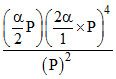 = 8P3α5
= 8P3α5
1 - α
KP =
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
In a closed container maintained at 1 atm pressure and 25o C, 2 moles of SO2(g) and 1 mole of O2(g) were allowed to reac...For the following gases equilibrium, N2O4 (g) ⇋ 2NO2 (g)Kp is found to be equal to Kc. This is attained when :...For the reaction: CuSO4.3H2O(s) $\rightleftharpoons$CuSO4.H2O(s) + 2H2O(g); ΔH = 3360 cal. The dissociation pressure is ...Select the reaction for which the equilibrium constant is written as [MX3]2 = Keq. [MX2]2 [X2]...Under what pressure must an equimolar mixture of Cl2 and PCl3 be placed at 250oC in order to obtain 75% conversion of PC...