JEE Main | 2014Chemical EquilibriumHard
Question
For the reaction SO2(g) + 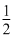 O2(g) ⇋ SO3(g), if KP = KC(RT)x where the symbols have usual meaning then the value of x is (assuming ideality)
O2(g) ⇋ SO3(g), if KP = KC(RT)x where the symbols have usual meaning then the value of x is (assuming ideality)
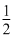 O2(g) ⇋ SO3(g), if KP = KC(RT)x where the symbols have usual meaning then the value of x is (assuming ideality)
O2(g) ⇋ SO3(g), if KP = KC(RT)x where the symbols have usual meaning then the value of x is (assuming ideality)Options
A.- 1
B.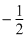
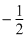
C.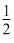
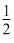
D.1
Solution
SO2(g) +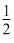 O2(g) ⇋ SO3(g)
O2(g) ⇋ SO3(g)
KP = KC(RT)x
x = ᐃng = no. no. of gaseous moles in product
- no. of gaseous moles in reactant
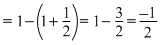
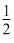 O2(g) ⇋ SO3(g)
O2(g) ⇋ SO3(g)KP = KC(RT)x
x = ᐃng = no. no. of gaseous moles in product
- no. of gaseous moles in reactant
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
Which of the following statements is correct for a reversible process in a state of equilibrium ?...The equilibrium constant of the following are : N2 + 3H2 ⇌ 2NH3            K1 N2 + O2 ⇌ 2NO      Â...The two equilibria AB$\rightleftharpoons$A+ + B– and AB + B– $\rightleftharpoons$AB2− are simultaneously maintained in a...In an evacuated rigid vessel of volume V litre, one mole of solid ammonium carbonate (NH2CONH4) is taken and the vessel ...For the reaction CaCO3(s) ⇋ CaO(s) + CO2(g), the pressure of CO2(g) depends on :...