Chemical EquilibriumHard
Question
The equation α = 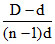 is correctly matched for :
is correctly matched for :
Options
A.A(g) ⇋ nB/2(g) + nC/3(g)
B.A(g) ⇋ nB/3(g) + (2n/3)C(g)
C.A(g) ⇋ (n/2)B(g) + (n/4)C(g)
D.A(g) ⇋ (n/2)B(g) + C(g)
Solution
We know that
d =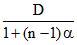
where n is number of mole of gases produced from one mole of a gas.
d =
where n is number of mole of gases produced from one mole of a gas.
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
The reaction which proceeds towards completion in the forward direction is...A 10 litre box contains O3 and O2 at equilibrium at 2000 K. KP = 4 × 1014 atm for 2O3 (g) ⇋ 3O2(g). Assume th...For dissociation of a gas N2O5 as N2O5(g) $\rightleftharpoons$2NO2(g) + ½ O2(g). The reaction is performed at constant t...Steam reacts with iron at high temperature to give hydrogen gas and Fe3O4(s). The corrent expression for the equilibrium...The equilibrium constant for the reaction: H3BO3 + Glycerin$\rightleftharpoons$ (H3BO3 + Glycerin complex) is 0.90. How ...