Chemical EquilibriumHard
Question
A 10 litre box contains O3 and O2 at equilibrium at 2000 K. KP = 4 × 1014 atm for 2O3 (g) ⇋ 3O2(g). Assume that PO2 > > PO3 and if total pressure is 8 atm, then partial pressure of O3 will be :
Options
A.8 × 10-5 atm
B.11.3 × 10-7 atm
C.9.71 × 10-6 atm
D.9.71 × 10-2 atm
Solution
2O3(g) ⇋ 3O2(g) Kp = 4 × 1014 pO2 > > pO3
Kp =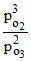 pO2 + PO3 = 8 ⇒ PO2 ≃ 8 atm.
pO2 + PO3 = 8 ⇒ PO2 ≃ 8 atm.
4 × 1014 =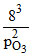 p2O3 = 11.3 × 10-7
p2O3 = 11.3 × 10-7
Kp =
4 × 1014 =
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
What is the equilibrium expression for the reaction P4(s) + 5O2(g) ⇋ P4O10(s) ?...Variation of log10 K with is shown by the following graph in which straight line is at 45o, hence ᐃHo is :...Ammonia gas at 15 atm is introduced in a rigid vessel at 300 K. At equilibrium the total pressure of the vessel is found...CuSO4.5H2O(s) $\rightleftharpoons$CuSO4.3H2O(s) + 2H2O(g), KP for this equilibrium is 1.0 × 10−4 atm2 at 25oC. What is t...In which of the following equilibrium Kc and Kp are not equal ?...