Chemical EquilibriumHard
Question
Equilibrium constant for the following equilibrium is given at 0ºC.
Na2HPO4 . 12H2O (s) ⇋ Na2HPO4 . 7H2O (s) + 5H2O(g) KP = 31.25 × 10-13
At equilibrium what will be partial pressure of water vapour :
Na2HPO4 . 12H2O (s) ⇋ Na2HPO4 . 7H2O (s) + 5H2O(g) KP = 31.25 × 10-13
At equilibrium what will be partial pressure of water vapour :
Options
A.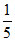 × 10-3 atm
× 10-3 atm
B.0.5 × 10-3 atm
C.5 × 10-2 atm
D.5 × 10-3 atm
Solution
Na2HPO4 .12H2O(s) ⇋ Na2HPO4.7H2O (s) + 5H2O (g) KP = 31.25 × 10-13
KP = (PH2O)5
(PH2O)5 = 31.25 × 10-13
(PH2O)5 = (3125)1/5 × (10-15)1/5
(PH2O)5 = 5 × 10-3
KP = (PH2O)5
(PH2O)5 = 31.25 × 10-13
(PH2O)5 = (3125)1/5 × (10-15)1/5
(PH2O)5 = 5 × 10-3
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
A vessel at 1000 K contains CO2 with a pressure of 0.5 atm. Some of the CO2 is converted into CO on the addition of grap...What is the minimum mass of CaCO3 (s), below which it decomposes completely, required to establish equilibrium in a 6.50...For the equilibrium SrCl2 · 6H2O(s) ⇋ SrCl2 · 2H2O(s) + 4H2O (g) the equilibrium constant KP = 16 × 10-12 atm...In an evacuated rigid vessel of volume V litre, one mole of solid ammonium carbonate (NH2CONH4) is taken and the vessel ...For a gaseous equilibrium 2A(g) $\rightleftharpoons$2B(g) + C(g), KP has a value 1.8 at 700 K. The value of KC for the e...