Chemical EquilibriumHard
Question
At 727oC and 1.23 atm of total equilibrium pressure, SO3 is partially dissociated into SO2 and O2 according to SO3(g) ⇋ SO2(g) + 1/2O2(g). The density of equilibrium mixture is 0.9 gm/litre. The degree of dissociation is :
Options
A.1/3
B.2/3
C.l/4
D.1/5
Solution
SO3(g) ⇋ SO2(g) + 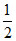 O2(g) Mmix =
O2(g) Mmix = 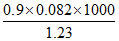 = 60
= 60
1 - α α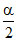 Mmix =
Mmix = 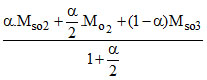
1 +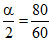
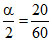
α =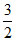
1 - α α
1 +
α =
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
The equilibrium constant for reaction H2(g) + CO2(g) ⇋ H2O(g) + CO(g) is 1·8 at 1000oC. If one mole of H2 and 1·0 ...Rate of disappearance of the reactant ‘A’ in the reversible reaction A$\rightleftharpoons$B at two temperatures is given...The first and second dissociation constants of an acid H2A are 1.0 × 10-5 and 5.0 × 10-10 respectively.The ove...Which one of the following statements is not correct?...For the reaction, 2NOBr(g) $\rightleftharpoons$2NO(g) + Br2(g), the ratio $\frac{K_{p}}{P}$, where P is the total pressu...