Chemical EquilibriumHard
Question
The equilibrium constant for reaction H2(g) + CO2(g) ⇋ H2O(g) + CO(g) is 1·8 at 1000oC. If one mole of H2 and 1·0 mole of CO2 are placed in a one litre flask. The final equilibrium concentration of CO at 1000oC will be 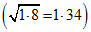 : -
: -
Options
A.0.290 M
B.0.386 M
C.0.573 M
D.0.688 M
Solution
H2(g) + CO2(g) ⇋ H2O(g) + CO(g)
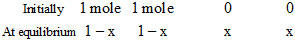
KC =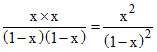
1.8 =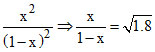 = 1.34
= 1.34
or x = 1.34 - 1.34 x
or x =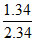 = 0.573 m
= 0.573 m
KC =
1.8 =
or x = 1.34 - 1.34 x
or x =
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
NH4HS(S) ⇋ NH3(g) + H2S(g)The equilibrium pressure at 25oC is 0.660 atm. What is KP for the reaction....Which of the following lines correctly show the temperature dependence of equilibrium constant, K, for an exothermic rea...For the reaction N2O4(g)$\rightleftharpoons$ 2NO2(g), the relation between the degree of dissociation of N2O4(g) at pres...Introduction of inert gas (at the same temperature) will affect the equilibrium if :...The equilibrium constant for the reaction CO(g) + H2O(g) $\rightleftharpoons$CO2(g) + H2(g) is 3.0 at 500 K. In a 2.0 L ...