Chemical EquilibriumHard
Question
X2(g) + Y2(g) ⇋ 2XY(g) reaction was studied at a certain temperature. In the beginning 1 mole of X2 was taken in a one litre flask and 2 moles of Y2 was taken in another 2 litre flask and both these containers are connected so equilibrium can be established. What is the equilibrium concentration of X2 and Y2? Given Equilibrium concentration of [XY] = 0.6 moles/litre.
Options
A.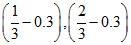
B.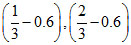
C.(1 - 0.3), (2 - 0.3)
D.(1 - 0.6), (2 - 0.6)
Solution
X2 + Y2 ⇋ 2XY
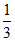 - x
- x 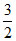 - x 2x 2x = 0.6 ⇒ x = 0.3
- x 2x 2x = 0.6 ⇒ x = 0.3
[x2] =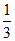 - 0.3 [y2] =
- 0.3 [y2] = 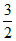 - 0.3
- 0.3
Therefore, (A) option is correct.
[x2] =
Therefore, (A) option is correct.
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
Equilibrium constant for the following equilibrium is given at 0ºC.Na2HPO4 . 12H2O (s) ⇋ Na2HPO4 . 7H2O (s) + 5H2O...For the system : P(s) ⇋ 2Q(g) + 3 R(g)keeping the temperature constant equilibrium is some how disturbed such that...In the system, LaCl3(s) + H2O(g) + heat$\rightleftharpoons$LaClO(s) + 2HCl(g), equilibrium is established. More water va...van 't Hoff equations show the effect of temperature on equilibrium constants KC and KP. KP and KC varies with temperatu...When a mixture of N2 and H2 in the volume ratio of 1: 5 is allowed to react at 700 K and 103 atm pressure, 0.4 mole frac...