Chemical EquilibriumHard
Question
For the system : P(s) ⇋ 2Q(g) + 3 R(g)
keeping the temperature constant equilibrium is some how disturbed such that at new equilibrium, concentration of R is increased by a factor of 2. At new equilibrium concentration of Q changes to-
keeping the temperature constant equilibrium is some how disturbed such that at new equilibrium, concentration of R is increased by a factor of 2. At new equilibrium concentration of Q changes to-
Options
A.2 times its original value
B.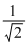 times its original value
times its original value
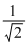 times its original value
times its original valueC.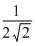 times its original value
times its original value
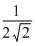 times its original value
times its original valueD.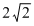 times its original value
times its original value
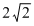 times its original value
times its original valueSolution
KC = [R]03 [Q]02
Temperature is constant hence Kc is also constant
At new eq.
Kc = [2R]3 [Q]2
Q′ =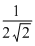 Q0
Q0
Temperature is constant hence Kc is also constant
At new eq.
Kc = [2R]3 [Q]2
Q′ =
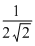 Q0
Q0Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
NH4HS(S) ⇋ NH3(g) + H2S(g)The equilibrium pressure at 25oC is 0.660 atm. What is KP for the reaction....A 20 litre container at 400 K contains CO2(g) at pressure 0.4 atm and an excess of SrO (neglect the volume of solid SrO)...For the given reaction at constant pressure, n A (g) ⇋ An (g)Initial moles 1 0Moles at equilibrium 1 - α ^...At 127°C and 1 atm pressure, PCl5(g) is partially dissociated into PCl2(g) and Cl2(g) as PCl5(g) $\rightleftharpoons$PCl...The equilibrium constant for the reaction SO3(g) ⇋ SO2(g) + O2(g) is Kc = 4.9 × 10-2. The value of Kc for the...