Chemical EquilibriumHard
Question
An equilibrium mixture in a vessel of capacity 100 litre contain 1 mol N2, 2 mol O2 and 3 mol NO. Number of moles of O2 to be added so that at new equilibrium the conc. of NO is found to be 0.04 mol/lit.:
Options
A.(101/18)
B.(101/9)
C.(202/9)
D.None of these
Solution
N2 (g) + O2 (g) 2NO (g)
1mole 2mole 3mole
KC =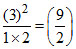 .
.
Let a mole of O2 is added, Then,
N2 (g) + O2 (g) 2NO (g)
1mole 2mole 3mole
t = 0 1 (2 + a) 3
(1 -x) (2 + a)-x (3 + 2x)
[NO] =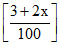 = 0.04 ; (3 + 2x) = 4.
= 0.04 ; (3 + 2x) = 4.
2x = 1, x = 0.5.
KC =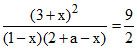
KC =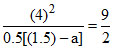
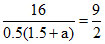
=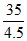 = [1.5 + a]
= [1.5 + a]
7.11 = 1.5 + a .
a =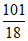 = 5.61
= 5.61
1mole 2mole 3mole
KC =
Let a mole of O2 is added, Then,
N2 (g) + O2 (g) 2NO (g)
1mole 2mole 3mole
t = 0 1 (2 + a) 3
(1 -x) (2 + a)-x (3 + 2x)
[NO] =
2x = 1, x = 0.5.
KC =
KC =
=
7.11 = 1.5 + a .
a =
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
For the reaction, A + 2B ⇋ 2C, the rate constants for the forward and the backward reactions are 1 × 10-4 and...For the reaction H2(g) + I2(g) $\rightleftharpoons$2HI(g), the value of equilibrium constant is 9.0. The degree of disso...The exothermic formation of ClF3 is represented by the equation: Cl2(g) + 3F2(g) ⇋ 2ClF3(g) ; ᐃrH = - 329 kJ...In the equilibrium mixture H2(g) + I2(g) $\rightleftharpoons$2HI(g), the mole ratio of gases are $\sqrt{2}:\sqrt{2}:10$,...If for 2A2B(g) ⇋ 2A2(g) + B2(g), Kp = TOTAL PRESSURE (at equilibrium) and starting the dissociation from 4 mol of ...