Chemical EquilibriumHard
Question
The exothermic formation of ClF3 is represented by the equation:
Cl2(g) + 3F2(g) ⇋ 2ClF3(g) ; ᐃrH = - 329 kJ
Which of the following will increase the quantity of ClF3 in an equilibrium mixture of Cl2, F2 and ClF3 ?
Cl2(g) + 3F2(g) ⇋ 2ClF3(g) ; ᐃrH = - 329 kJ
Which of the following will increase the quantity of ClF3 in an equilibrium mixture of Cl2, F2 and ClF3 ?
Options
A.Increasing the temperature
B.Removing Cl2
C.Increasing the volume of the container
D.Adding F2
More Chemical Equilibrium Questions
For the reaction NH2COONH4(s) $\rightleftharpoons$2NH3(g) + CO2(g), KP = 3.2 × 10−5 atm3. The total pressure of the gase...Which of the following lines correctly show the temperature dependence of equilibrium constant, K, for an exothermic rea...In which of the following equilibrium Kc and Kp are not equal ?...The dissociation of ammonium carbamate may be represented by the equation NH4CO2NH2(s) ⇋ 2NH3(g) + CO2(g)ᐃH0...In the reaction C(s) + CO2(g) ⇋ 2CO(g) the equilibrium pressure is 12 atm. If 50% of CO2 reacts than Kp will be :...
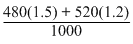 = 1.344M
= 1.344M