Chemical EquilibriumHard
Question
If for 2A2B(g) ⇋ 2A2(g) + B2(g), Kp = TOTAL PRESSURE (at equilibrium) and starting the dissociation from 4 mol of A2B then :
Options
A.degree of dissociation of A2B will be (2/3).
B.total no. of moles at equilibrium will be (14/3).
C.at equilibrium the no. of moles of A2B are not equal to the no. of moles of B2.
D.at equilibrium the no. of moles of A2B are equal to the no. of moles of A2.
Solution
2A2B(g) ⇋ 2A2(g) + B2(g) KP = P
4(1 - α) 4α 2α ∑ n = 4 + 2α
KP =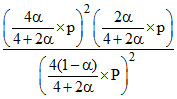 = P ⇒ 2α3 = (1 - α)2 (4 + 2α)
= P ⇒ 2α3 = (1 - α)2 (4 + 2α)
2α3 = (1 + α2 - 2α) (4 + 2α)
2α3 = 4 + 2α + 4α2 + 2α3 - 8α - 4α2
α =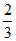
4(1 - α) 4α 2α ∑ n = 4 + 2α
KP =
2α3 = (1 + α2 - 2α) (4 + 2α)
2α3 = 4 + 2α + 4α2 + 2α3 - 8α - 4α2
α =
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
The equilibrium NH4HS(s) $\rightleftharpoons$NH3(g) + H2S(g) is achieved at the equilibrium pressure of ‘X’ bar at T K. ...The position of equilibrium will shift in the given direction by the addition of inert gas at constant pressure in which...In an evacuated vessel of capacity 112 L, 4 moles of Ar(g) and 5 moles of PCl5(g) were introduced and the temperature is...The two equilibria AB$\rightleftharpoons$A+ + B– and AB + B– $\rightleftharpoons$AB2− are simultaneously maintained in a...A cylinder fitted with a movable piston contains liquid water in equilibrium with water vapour at 25°C. Which of the fol...