Chemical EquilibriumHard
Question
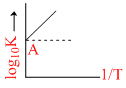
Variation of log10 K with 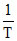 is shown by the following graph in which straight line is at 45o, hence ᐃHo is :
is shown by the following graph in which straight line is at 45o, hence ᐃHo is :

Options
A.+ 4.606 cal
B.- 4.606 cal
C.2 cal
D.- 2 cal
Solution
K = A eᐃH/RT
log K = log A -
log K = log A -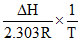
log K =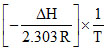 + log A.
+ log A.
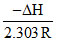 = 1.
= 1.
ᐃH = - 2.303 R = - 4.606 cal.
log K = log A -
log K = log A -
log K =
ᐃH = - 2.303 R = - 4.606 cal.
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
At 200°C, PCl5 dissociates as PCl5(g) $\rightleftharpoons$PCl3(g) + Cl2(g). It was found that the equilibrium vapours ar...For the reaction N2O4 (g) ⇋ 2NO2 (g), if percentage dissociation of N2O4 are 20%, 45%, 65% & 80%, then the sequenc...A quantity of 60 g of CH3COOH and 46 g of CH3CH2OH reacts in 5 L flask to form 44 g of CH3COOC2H5 at equilibrium. On tak...Solubility of a solute in a solvent (say, water) is dependent on the temperature as given by S = A.e–ΔH/RT, where ΔH is ...At constant temperature, the equilibrium constant (Kp)for the decomposition reaction,N2O4 ⇋ 2NO2 is expressed by K...