Chemical EquilibriumHard
Question
For the reaction N2O4 (g) ⇋ 2NO2 (g), if percentage dissociation of N2O4 are 20%, 45%, 65% & 80%, then the sequence of observed vapour densities will be :
Options
A.d20 > d45 > d65 > d80
B.d80 > d65 > d45 > d20
C.d20 = d45 = d65 = d80
D.(d20 = d45) > ( d65 = d80)
Solution
(VD)mix = 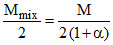
so, as α increases (VD)mix decreases.
so, as α increases (VD)mix decreases.
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
The equilibrium constant for the reaction N2(g) + O2(g) $\rightleftharpoons$2NO(g) is 0.09 at 3500 K. The fraction of eq...The formation constant of Ni(NH3)62+ is 6 × 108 at 250C. If 50 ml of 2.0 M NH3 is added to 50 ml of 0.20 M solution...For the reaction X2(g) + Y2(g) $\rightleftharpoons$2XY(g), 2 moles of ‘X2’ was taken in a 2 L vessel and 3 moles of ‘Y2’...What is the approximate value of log KP for the following reaction?N2(g) + 3H2(g) $\rightleftharpoons$ 2NH3(g) at 25°C. ...An equilibrium mixture in a vessel of capacity 100 litre contain 1 mol N2, 2 mol O2 and 3 mol NO. Number of moles of O2 ...