Chemical EquilibriumHard
Question
At constant temperature, the equilibrium constant (Kp)for the decomposition reaction,N2O4 ⇋ 2NO2 is expressed by Kp = 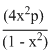 , where p = pressure, x = extent of decomposition. Which one of the following statement is true ?
, where p = pressure, x = extent of decomposition. Which one of the following statement is true ?
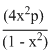 , where p = pressure, x = extent of decomposition. Which one of the following statement is true ?
, where p = pressure, x = extent of decomposition. Which one of the following statement is true ? Options
A.Kp increases with incrase of p
B.Kp increases with incrase of x
C.Kp increases with decrease of x
D.Kp remains constant with change in p and x
Solution
Kp is a characteristic constant for a reaction and changes with temperature only.
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
The following reaction has an equilibrium constant KC equal to 3.07 × 10–4 at 24°C....The correct relationship between free energy change in a reaction and the corresponding equilibrium constant Kc is...For the reaction, CO(g) + Cl2(g) ⇋ COCl2(g) the is equal to...The two equilibria AB$\rightleftharpoons$A+ + B– and AB + B– $\rightleftharpoons$AB2− are simultaneously maintained in a...Equilibrium constants for some reactions are given. In which of the following case does the reaction go farthest to comp...