Chemical EquilibriumHard
Question
Sulfide ion in alkaline solution reacts with solid sulfur to form polysulfide ions having formulae S22-, S32-, S42- and so on. The equilibrium constant for the formation of S22- is 12 (K1) & for the formation of S32- is 132 (K2), both from S and S2-. What is the equilibrium constant for the formation of S32- from S22- and S ?
Options
A.11
B.12
C.132
D.None of these
Solution
S (s) + S2- (aq) ⇋ S22- (aq) K1 = 12
2S (s) + S2- (aq) ⇋ S32- (aq) K2 = 132
Now , S22- (aq) + S (s) S32- (aq) Keq =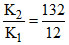 = 11
= 11
2S (s) + S2- (aq) ⇋ S32- (aq) K2 = 132
Now , S22- (aq) + S (s) S32- (aq) Keq =
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
The equilibrium constant Kp1 and Kp2 for the reactions A ⇋ 2B and P ⇋ Q + R, respectively, are in the ratio ...For a reaction,A → B ; if log10 K(sec-1) = 14 - the Arrheniums parameter and energy of activation for the reaction...In a closed tube, HI(g) is heated at 440°C up to establishment of equilibrium. If it dissociates into H2(g) and I2(g) up...In the decomposition equilibrium of a certain alkaline earth metal carbonate, the partial pressure of CO2 becomes a hund...When α-D-glucose is dissolved in water, it undergoes a partial conversion to β-D-glucose. This conversion is called muta...