Chemical EquilibriumHard
Question
The equilibrium constant Kp1 and Kp2 for the reactions A ⇋ 2B and P ⇋ Q + R, respectively, are in the ratio of 2 : 3. If the degree of dissociation of A and P are equal, the total pressure at equilibrium is,
Options
A.1 : 36
B.1 : 9
C.1 : 6
D.1 : 4
Solution
For A ⇋ 2B
Initial 1 0
At equilibrium 1 - α 2α
Total moles = 1 - α + 2α = 1 + α
∴ PA = (mole fraction of A) × Initial pressure =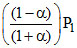
Similarly PB =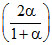
KP1 =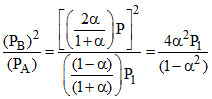
For P ⇋ Q + R
Initial 1 0 0
At equilibrium 1 - α α α
Total moles = 1 - α + α + α = 1 + α
∴ PP =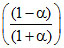 P2 ; PQ =
P2 ; PQ = 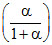 P2; PR =
P2; PR = 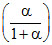 P2
P2
KP2 =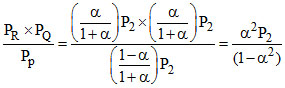
∴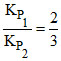 (given) =
(given) = 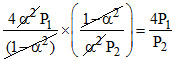
∴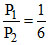
Initial 1 0
At equilibrium 1 - α 2α
Total moles = 1 - α + 2α = 1 + α
∴ PA = (mole fraction of A) × Initial pressure =
Similarly PB =
KP1 =
For P ⇋ Q + R
Initial 1 0 0
At equilibrium 1 - α α α
Total moles = 1 - α + α + α = 1 + α
∴ PP =
KP2 =
∴
∴
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
CuSO4.5H2O(s) $\rightleftharpoons$CuSO4.3H2O(s) + 2H2O(g), KP for this equilibrium is 1.0 × 10−4 atm2 at 25oC. What is t...Which of the following statements is correct for a reversible process in a state of equilibrium ?...From the following data, (i) H2(g) + CO2(g) $\rightleftharpoons$ H2O(g) + CO(g); K2000 K = 4.4(ii) 2H2O(g) $\rightleftha...One mole of N2O4 (g) at 300 K is kept in a closed container under one atm. It is heated to 600 K when 20% by mass of N2O...A vessel at 1000 K contains CO2 with a pressure of 0.5 atm. Some of the CO2 is converted into CO on the addition of grap...