Chemical EquilibriumHard
Question
When alcohol (C2H5OH (l)) and acetic acid (CH3COOH (l)) are mixed together in equimolar ratio at 27oC , 33% of each is converted into ester. Then the KC for the equilibrium
C2H5OH(l) + CH3COOH (l) ⇋ CH3COOC2H5 (l) + H2O(l).
C2H5OH(l) + CH3COOH (l) ⇋ CH3COOC2H5 (l) + H2O(l).
Options
A.4
B.1/4
C.9
D.1/9
Solution
C2H5OH (l) + CH3COOH (l) ⇋ CH3COOC2H5 (l) + H2O (l).
a a 0 0
a 0.33a a - 0.33a 0.33a 0.33a
KC =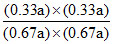 = KC = 1/4.
= KC = 1/4.
a a 0 0
a 0.33a a - 0.33a 0.33a 0.33a
KC =
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
Which one of the following has lowest melting point :-...Steam decomposes at high temperature according to the following equation.2H2O(g) $\rightleftharpoons$2H2(g) + O2(g); ΔHo...In a container equilibrium N2O4 (g) ⇋ 2NO2 (g) is attained at 25oC. The total equilibrium pressure in container is...Equilibrium constant for the reactions, 2 NO + O2 ⇋ 2 NO2 is KC1 ; NO2 + SO2 ⇋ SO3 + NO is KC2 and 2 SO3 ...At a certain temperature, the value of KP for the reaction: 2CO(g) $\rightleftharpoons$CO2(g) + C(graphite) is 0.1 atm−1...