Chemical EquilibriumHard
Question
In a container equilibrium N2O4 (g) ⇋ 2NO2 (g)
is attained at 25oC. The total equilibrium pressure in container is 380 torr. If equilibrium constant of above equilibrium is 0.667 atm, then degree of dissociation of N2O4 at this temperature will be :
is attained at 25oC. The total equilibrium pressure in container is 380 torr. If equilibrium constant of above equilibrium is 0.667 atm, then degree of dissociation of N2O4 at this temperature will be :
Options
A.1/3
B.1/2
C.2/3
D.1/4
Solution
KP = 0.667 atm = 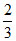 atm =
atm = 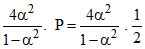
So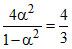 ⇒ 3α2 = 1 - α2
⇒ 3α2 = 1 - α2
So α2 =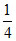 ⇒ α =
⇒ α = 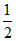
So
So α2 =
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
In the reaction N2(g) + 3H2(g) $\rightleftharpoons$2NH3(g) + Heat. One mole of N2 reacts with three moles of H2. If at e...PCl5 (molecular mass = M) dissociates into PCl3 and Cl2 as PCl5(g) $\rightleftharpoons$PCl3(g) + Cl2(g). If the total pr...Iron fillings and water were placed in a 5 L vessel and sealed. The tank was heated to 1000oC. Upon analysis, the tank w...An aqueous solution contains 0.10 M H2S and 0.20 M HCl. If the equilibrium constants for the formation of HS- from H2S i...Listed in the table are forward and reverse rate constant for the reaction 2NO(g) ⇋ N2(g) + O2(g) Select the corre...