Chemical EquilibriumHard
Question
The equilibrium constant of the reaction SO2(g) + 1/2O2(g) ⇋ SO3(g) is 4 × 10-3 atm-1/2. The equilibrium constant of the reaction 2SO3(g) ⇋ 2SO2(g) + O2(g) would be :
Options
A.250 atm
B.4 × 103 atm
C.0.25 × 104 atm
D.6.25 × 104 atm
Solution
SO2(g) + 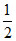 O2(g) ⇋ SO3(g) Kp = 4 × 10-3
O2(g) ⇋ SO3(g) Kp = 4 × 10-3
SO3 ⇋ SO2(g) +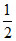 O2(g) K′p =
O2(g) K′p = 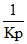
K′p =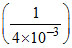
2SO3 ⇋ 2SO2 + O2(g)
K″p = (K′p)2 =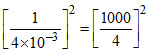 = 6250 = 625×102 6.25 × 104 atm.
= 6250 = 625×102 6.25 × 104 atm.
SO3 ⇋ SO2(g) +
K′p =
2SO3 ⇋ 2SO2 + O2(g)
K″p = (K′p)2 =
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
At T K, a compound AB2(g) dissociates according to the reaction 2AB2(g) $\rightleftharpoons$2AB(g) + B2(g), with a degre...For the reaction 2HI(g) ⇋ H2(g) + I2(g) the degree of dissociation (α) of HI(g) is related to equilibrium con...Consider the following equilibrium in a closed container N2O4(g) ⇋ 2NO2(g) At a fixed temperature, the volume of t...The Haber’s process for the manufacture of ammonia is usually carried out at about 500oC. If a temperature of about 250o...For the reaction CO(g) + H2O(g) ⇋ CO2(g) + H2(g) at a given temperature the equilibrium amount of CO2 (g) can be i...