Chemical EquilibriumHard
Question
For the reaction 2HI(g) ⇋ H2(g) + I2(g) the degree of dissociation (α) of HI(g) is related to equilibrium constant KP by the expression :-
Options
A.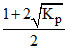
B.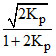
C.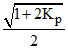
D.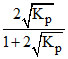
More Chemical Equilibrium Questions
In a system A(s) $\rightleftharpoons$2B(g) + 3C(g), if the concentration of C at equilibrium is increased by a factor of...What is the minimum mass of CaCO3 (s), below which it decomposes completely, required to establish equilibrium in a 6.50...The standard emf of cell, involving one electron change is found to be 0.591 V at 25ºC. The equilibrium constant of the ...An equilibrium mixture in a vessel of capacity 100 litre contain 1 mol N2, 2 mol O2 and 3 mol NO. Number of moles of O2 ...For the reaction CaCO3(s) $\rightleftharpoons$CaO(s) + CO2(g), the value of KP is...