Atomic StructureHard
Question
If the nitrogen atom had electronic configuration 1s7, it word have energy lower than that of the normal ground state congiguration ls22s22s3, because the electrons would be closer to the nucleus, yet 1s7 is not obsrrved because it violates :
Options
A.Heisenderg uncertainty principle
B.Hund′s rule
C.Pauli′s exclusion principle
D.Bohr′s postulate of stationary orbits
More Atomic Structure Questions
An electron and a proton are accelerated through a potential V. If Pe and Pp are their momentum, then PP: Pe ratio is ap...An α–particle is accelerated from rest through a potential difference of 6.0V. Its de Broglie wavelength is...When the reaction, 2NO2(g) ⇋ N2O4(g) reaches equilibrium at 298 K. The partial of NO2 and N2O4 are 0.2 KPa and 0.4...The ratio of circumference of third and second orbits of He+ ion is...Photons of equal energy were incident on two different gas samples. One sample containing H-atoms in the ground state an...
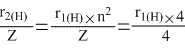 = r1(H)
= r1(H)