Atomic StructureHard
Question
When the reaction, 2NO2(g) ⇋ N2O4(g) reaches equilibrium at 298 K. The partial of NO2 and N2O4 are 0.2 KPa and 0.4 KPa, respectively. What is the equilibrium constant Kp of the above reaction at 298 K ?
Options
A.0.1
B.0.5
C.1.0
D.2
Solution
2NO2 ⇋ N2O4
At equilibrium 0.2 kPa 0.4 kPa
Kp =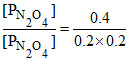 = 10
= 10
At equilibrium 0.2 kPa 0.4 kPa
Kp =
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
The neon atoms has a radius of 160 pm. What is the edge of the unit cell of a face centered structure of neon ?...In which transition, one quantum of energy is emitted ?...Incorrect statement is :-...A rod PQ of mass M and length L is hinged at end P. The rod is kept horizontal by a massless string tied to point Q as s...In a sample of H-atoms, electrons de-excite from a level ′n′ to 1. The total number of lines belonging to Ba...