Atomic StructureHard
Question
Photons of equal energy were incident on two different gas samples. One sample containing H-atoms in the ground state and the other sample containing H-atoms in some excited state with a principal quantum number ′n′. The photonic beams totally ionise the H-atoms. If the difference in the kinetic energy of the ejected electrons in the two different cases is 12.75 eV. Then find the principal quantum number ′n′ of the excited state.
Options
A.1
B.2
C.3
D.4
Solution
KE1 = Ephoton – BEn = 1
KE2 = Ephoton – BEn = n
KE2 - KE1 = BEn = 1 - BEn = n = 13.6 Z2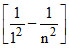 = 12.75 (given).
= 12.75 (given).
∴ n2 = 16 or n = 4.
BE : Binding energy.
KE2 = Ephoton – BEn = n
KE2 - KE1 = BEn = 1 - BEn = n = 13.6 Z2
∴ n2 = 16 or n = 4.
BE : Binding energy.
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
The wavelength of visible light is:...The configuration [Ar] 3d10 4s2 4p4 is similar to that of...When an electron jumps from the second orbit to fourth orbit, its distance from nucleus increases by 2.116 Å. The atom o...In which transition, one quantum of energy is emitted ?...When alpha particles are sent through a thin metal foil, most of them go straight through the foil because :...