Atomic StructureHard
Question
Which onr of the following sets of quantum numbers represents an impossible arrangement ?
Options
A.

B.

C.

D.

Solution
For the given value of n, m cannot have value + n. It may be upto 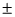 (n - 1) only Thus for n = 3, m can′t be 3.
(n - 1) only Thus for n = 3, m can′t be 3.
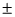 (n - 1) only Thus for n = 3, m can′t be 3.
(n - 1) only Thus for n = 3, m can′t be 3. Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
According to Bohr′s theory, the angular momentum of an electron in 5th orbit is...The uncertainty in position and velocity of the particle are 0.1 nm and 5.27 × 10-27 ms-1 respectively then the mas...A certain dye absorbs light of l = 4000 and then fluorescence light of 5000 . Assuming that under given conditions 50% o...What possibly can be the ratio of the de Broglie wavelengths for two electrons each having zero initial energy and accel...Magnetic moment of Xn+ (Z = 26) is B.M. Hence number of unpaired electrons and value of n respectively are ;...