Atomic StructureHard
Question
The uncertainty in position and velocity of the particle are 0.1 nm and 5.27 × 10-27 ms-1 respectively then the mass of the particle is : (h = 6.625 × 10-34Js)
Options
A.200 g
B.300 g
C.100 g
D.1000 g
Solution
ᐃx = 0.1 × 10-9 m.
DV = 5.27 × 10-27 ms-1.
ᐃV = 5.27 × 10-27 ms-1.
∴ ᐃx × mᐃV =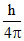
∴ 0.1 × 10-9 × m × 5.27 × 10-27 = 0.527 × 10-34.
∴ m = 0.1 kg. = 100 gm.
DV = 5.27 × 10-27 ms-1.
ᐃV = 5.27 × 10-27 ms-1.
∴ ᐃx × mᐃV =
∴ 0.1 × 10-9 × m × 5.27 × 10-27 = 0.527 × 10-34.
∴ m = 0.1 kg. = 100 gm.
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
In H-atom, the wave number ratio is 108: 7 is for...Photoelectric emission is observed from a metal surface for frequencies v1 and v2 of the incident radiation (v1 > v2)...The specific charge of cathode rays...In the absence of external magnetic field, d-orbital is...In different experiments, α-particles, proton, deuteron and neutron are projected towards gold nucleus with the same ki...