Atomic StructureHard
Question
Magnetic moment of Xn+ (Z = 26) is 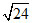 B.M. Hence number of unpaired electrons and value of n respectively are ;
B.M. Hence number of unpaired electrons and value of n respectively are ;
Options
A.Magnetic moment of Xn+ (Z = 26) is 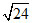 B.M. Hence number of unpaired electrons and value of n respectively are ;
B.M. Hence number of unpaired electrons and value of n respectively are ;
B.2, 4
C.3, 1
D.0, 2
Solution
Magnetic moment = 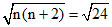 B.M
B.M
No. of unpaired electron = 4.
X26 : 1s2 2s22p63s23p63d64s2.
To get 4 unpaired electrons, outermost configuration will be 3d6.
∴ No. of electrons lost = 2 (from 4s2).
∴ n = 2.
No. of unpaired electron = 4.
X26 : 1s2 2s22p63s23p63d64s2.
To get 4 unpaired electrons, outermost configuration will be 3d6.
∴ No. of electrons lost = 2 (from 4s2).
∴ n = 2.
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
A sample of a hydrate of barium chloride weighing 61 g was heated until all the water of hydration is removed. The dried...Electron moving with a velocity of ′V′ has a certain value of de-Broglie wave length. The velocity of neutro...The energy of an electron in the first Bohr orbit of H-atom is -13.6eV. The possible energy value (s) of the excited sta...The orbital angular momentum of an electron in 2s orbital is :...In a H-like sample, electrons make transition from 4th excited state to 2nd state then...