Atomic StructureHard
Question
A certain dye absorbs light of l = 4000 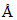 and then fluorescence light of 5000
and then fluorescence light of 5000 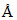 . Assuming that under given conditions 50% of the absorbed energy is re-emitted out as fluorescence, calculate the ratio of number of quanta emitted out to the number of quanta absorbed.
. Assuming that under given conditions 50% of the absorbed energy is re-emitted out as fluorescence, calculate the ratio of number of quanta emitted out to the number of quanta absorbed.
Options
A.5/8
B.8/5
C.3/8
D.8/3
Solution
Eemitted = 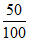 × No. of absorbed photon × Energy of absorbed photon.
× No. of absorbed photon × Energy of absorbed photon.
∴ ne ×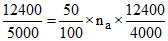
∴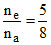 .
.
∴ ne ×
∴
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
Orbitals used in hybridisation of anion of CsBr3 is:-...If the total energy of an electron in hydrogen like atom in an excited state is -3.4 eV, then the de-Broglie wavelength ...In a H-like sample, electrons make transition from 4th excited state to 2nd state then...An ion Mna+ has the magnetic moment equal to 4.9 BM. The value of a is : (atomic no. of Mn = 25)...Neon does not form clathrate compound because -...