Chemical EquilibriumHard
Question
For the following gases equilibrium, N2O4 (g) ⇋ 2NO2 (g)
Kp is found to be equal to Kc. This is attained when :
Kp is found to be equal to Kc. This is attained when :
Options
A.0oC
B.273 K
C.1 K
D.12.19 K
Solution
KP = KC (RT)ᐃng = KC (RT)
so T =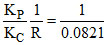 = 12.18 K
= 12.18 K
so T =
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
For the equilibium CH3 - CH2 - CH2 - CH3(g) ⇋ (g) equilibrium constant is found to be 1.732 at 298 K. Now if in a ...2 mole each of SO3, CO, SO2 and CO2 is taken in a one lit. vessel. If KC for SO3(g) + CO(g) ⇋ SO2(g) + CO2(g) is 1...A gaseous mixture contains 0.30 moles of CO, 0.10 moles of H2, and 0.03 moles of H2O vapour and an unknown amount of CH4...For the equilibrium LiCl·3NH3(s) $\rightleftharpoons$LiCl·NH3(s) + 2NH3(g); KP = 9 atm2 at 27°C. A 8.21 L vessel contain...In the following equilibrium N2O4(g) $\rightleftharpoons$2NO2(g), when 5 moles of each is taken and the temperature is k...