Chemical EquilibriumHard
Question
For the equilibium CH3 - CH2 - CH2 - CH3(g) ⇋ 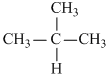 (g) equilibrium constant is found to be 1.732 at 298 K. Now if in a vessel at 298 K, a mixture of these two gases be taken as represented by the point P in the figure, predict what will happen
(g) equilibrium constant is found to be 1.732 at 298 K. Now if in a vessel at 298 K, a mixture of these two gases be taken as represented by the point P in the figure, predict what will happen
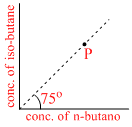
 (g) equilibrium constant is found to be 1.732 at 298 K. Now if in a vessel at 298 K, a mixture of these two gases be taken as represented by the point P in the figure, predict what will happen
(g) equilibrium constant is found to be 1.732 at 298 K. Now if in a vessel at 298 K, a mixture of these two gases be taken as represented by the point P in the figure, predict what will happen
Options
A.Immediately, above equilibrium will be setup
B.Above reaction will go in the forward direction till it attains equilibrium
C.Above reaction will go in the backward direction till it attains equilibrium
D.Nothing can be said
Solution
From given information, Q = tan 75o = 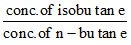 > Kc = √3
> Kc = √3
so backward reaction
so backward reaction
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
A certain weak acid has Ka = 1 × 10-4 . Calculate the equilibrium constant for its rx with a strong base :...In a flask, colourless N2O4 is in equilibrium with brown coloured NO2. At equilibrium, when the flask is heated at 100oC...For the reaction X2(g) + Y2(g) $\rightleftharpoons$2XY(g), 2 moles of ‘X2’ was taken in a 2 L vessel and 3 moles of ‘Y2’...Consider the following liquid - vapour equilibrium. Liquid ⇌ Vapour Which of the following relations is correct?...The equilibrium SOCl2(g) $\rightleftharpoons$SO2(g) + Cl2(g) is attained at 25oC in a closed rigid container and helium ...