Chemical EquilibriumHard
Question
2 mole each of SO3, CO, SO2 and CO2 is taken in a one lit. vessel. If KC for
SO3(g) + CO(g) ⇋ SO2(g) + CO2(g) is 1/9 then
SO3(g) + CO(g) ⇋ SO2(g) + CO2(g) is 1/9 then
Options
A.total no. of moles at equilibrium are less than 8
B.n(SO3) + n(CO2) = 4
C.[n(SO2)/n(CO)] < 1
D.both (B) and (C).
Solution
SO3(g) + CO(g) SO2 (g) + CO2 (g) 9 = 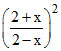
2 2 2 2
2 + x 2+x 2-x 2-x x = 1
neq = 3 + 3 + 1 + 1 = 8 n(SO3) + n(CO2) = 4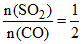 < 1
< 1
Therefore, (D) option is correct.
2 2 2 2
2 + x 2+x 2-x 2-x x = 1
neq = 3 + 3 + 1 + 1 = 8 n(SO3) + n(CO2) = 4
Therefore, (D) option is correct.
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
The value of KP for the reaction: H2(g) + I2(s) $\rightleftharpoons$2HI(g) is 6.4 × 10–4 atm. On close observation, it i...Some quantity of water is contained in a container as shown in figure. As neon is added to this system at constant press...At chemical equilibrium, the...In the decomposition equilibrium of a certain alkaline earth metal carbonate, the partial pressure of CO2 becomes a hund...In the system, LaCl3(s) + H2O(g) + heat ⇋ LaClO(s) + 2HCl(g), equilibrium is established. More water vapour is add...