Chemical EquilibriumHard
Question
The equilibrium N2 (g) + O2 (g) ⇋ 2NO (g) is estabilished in a reaction vessel of 2.5 L capacity. The amounts of N2 and O2 taken at the start were respectively 2 moles and 4 moles. Half a mole of nitrogen has been used up at equilibrium. The molar concentration of nitric oxide is :
Options
A.0.2
B.0.4
C.0.6
D.0.1
Solution
N2 + O2 ⇋ 2NO
Initial 2 moles 4moles
At Eq. 2 -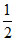 4 -
4 - 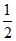 2 ×
2 × 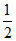 = 1 mol
= 1 mol
Molar concentration of NO at equilibrium =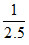 = 0.4
= 0.4
Initial 2 moles 4moles
At Eq. 2 -
Molar concentration of NO at equilibrium =
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
Methanol (CH3OH) can be prepared from CO and H2 as CO(g) + 2H2(g) $\rightleftharpoons$CH3OH(g); KP = 6.23 × 10−3 at 500 ...In a closed tube, HI(g) is heated at 440°C up to establishment of equilibrium. If it dissociates into H2(g) and I2(g) up...For the reaction, 2NOBr(g) $\rightleftharpoons$2NO(g) + Br2(g), the ratio $\frac{K_{p}}{P}$, where P is the total pressu...For the equilibrium 2SO2(g) + O2(g) ⇋ 2SO3(g), ᐃH = - 198 kJ, the equilibrium concentration of SO3 will be a...The amounts of 0.8 mol of PCl5 and 0.2 mole of PCl3 are mixed in a 1 L flask. At equilibrium, 0.4 mole of PCl3 is presen...