Chemical EquilibriumHard
Question
Given that the equilibrium constant for the reaction
2SO2(g) + O2(g) ⇋ 2SO3(g)
has a value of 278 at a particular temperature. What is the value of the equilibrium constant for the following reaction at the same temperature.
SO3(g) ⇋ SO2(g) + 1/2 O2(g)
2SO2(g) + O2(g) ⇋ 2SO3(g)
has a value of 278 at a particular temperature. What is the value of the equilibrium constant for the following reaction at the same temperature.
SO3(g) ⇋ SO2(g) + 1/2 O2(g)
Options
A.1.3 × 10-5
B.1.8 × 10-3
C.1.6 × 10-3
D.6.0 × 10-2
Solution
2SO2 + O2 ⇋ 2SO3
Keq = 278
SO3 ⇋ SO2 + 1/2 O2
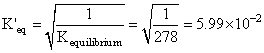
Keq = 278
SO3 ⇋ SO2 + 1/2 O2
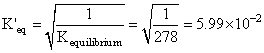
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
The equilibrium constant of the following are : N2 + 3H2 ⇌ 2NH3            K1 N2 + O2 ⇌ 2NO      Â...When calcium acetate is dissolved in water, heat is evolved. If the temperature is raised, then the solubility of calciu...NH4HS(S) ⇋ NH3(g) + H2S(g)The equilibrium pressure at 25oC is 0.660 atm. What is KP for the reaction....One mole of N2O4 (g) at 300 K is kept in a closed container under one atm. It is heated to 600 K when 20% by mass of N2O...For the given reaction at constant pressure, n A (g) ⇋ An (g)Initial moles 1 0Moles at equilibrium 1 - α ^...