SolutionHard
Question
When 100 ml of 0.2M BaCl2 (aq) is added to 100ml of 0.2M K2SO4 solution, of K2SO4 solution is lowered. Assume 100% dissociation of soluble salts and 100% precipitation of insoluble ones.
Options
A.Freezing point
B.Boiling point
C.Vapour pressure
D.cc
Solution
K2SO4 + BaCl2 → BaSO4 + 2KCl
0.02 0.02 0 0
0 0 0.02 0.04
ᐃTb = m Kbi
(ᐃTb)i = (0.02 × 2 + 0.02) × Kb = 0.06 Kb
(ᐃTb)final = 0.04 × 2 × Kb = 0.08 Kb
ᐃTb ↑ So B.P. ↑
(ᐃTf)i = - 0.06 Kf ; (ᐃTf)f = 0.08 Kf
ᐃTf ↑ So F.P. ↓
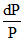 = Xsolute ; Xsolute ↑ So dP ↓ VP ↓
= Xsolute ; Xsolute ↑ So dP ↓ VP ↓
πinitial = M R T i = 0.06 RT
πfinal = 0.08 RT
So, π↑
0.02 0.02 0 0
0 0 0.02 0.04
ᐃTb = m Kbi
(ᐃTb)i = (0.02 × 2 + 0.02) × Kb = 0.06 Kb
(ᐃTb)final = 0.04 × 2 × Kb = 0.08 Kb
ᐃTb ↑ So B.P. ↑
(ᐃTf)i = - 0.06 Kf ; (ᐃTf)f = 0.08 Kf
ᐃTf ↑ So F.P. ↓
πinitial = M R T i = 0.06 RT
πfinal = 0.08 RT
So, π↑
Create a free account to view solution
View Solution FreeMore Solution Questions
3.24 g of Hg(NO3)2 (molar mass = 324) dissolved in 1000 g of water constitutes a solution having a freezing point of - 0...Mole fraction of C3H5(OH)3 in a solution of 36 g of water and 46 g of glycerine is :...When only a little quantity of HgCl2(s) is added to excess KI(aq) to obtain a clear solution, which of the following is ...Y gm of non-volatile organic substance of molecular mass M is dissolved in 250 gm benzene. Molal elevation constant of b...An ideal mixture of liquids A and B with 2 moles of A and 2 moles of B has a total vapour pressure of 1 atm at a certain...