SolutionHard
Question
3.24 g of Hg(NO3)2 (molar mass = 324) dissolved in 1000 g of water constitutes a solution having a freezing point of - 0.0558oC while 21.68 g of HgCl2 (molar mass = 271) in 2000 g of water constitutes a solution with a freezing point of - 0.0744oC. The Kf for water is 1.86 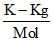 . About the state of ionization of these two solids in water it can be inferred that :
. About the state of ionization of these two solids in water it can be inferred that :
Options
A.Hg(NO3)2 and HgCl2 both are completely ionized
B.Hg(NO3)2 is fully ionized but HgCl2 is fully unionized
C.Hg(NO3)2 and HgCl2 both are completely unionized
D.Hg(NO3)2 is fully unionized but HgCl2 is fully ionized
Solution
0.0558 = i × 1.86 × 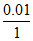 ⇒ i = 3
⇒ i = 3
⇒ fully ionized
0.0744 = i × 1.86 ×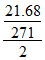 ⇒ i = 1
⇒ i = 1
⇒ fully unionized
⇒ fully ionized
0.0744 = i × 1.86 ×
⇒ fully unionized
Create a free account to view solution
View Solution FreeMore Solution Questions
Under ambient conditions, which among the following surfactants will form micelles in aqueous solution at lowest molar c...Which statement comparing solutions with pure solvent is not correct...Pressure cooker reduces cooking time because...Consider a binary mixture of volatile liquids. If at XA = 0.4 the vapour pressure of solution is 580 torr then the mixtu...Depression of freezing point of 0.01 molal aq. CH3COOH solution is 0.02046o. 1 molal urea solution freezes at - 1.86°C. ...