SolutionHard
Question
Mole fraction of C3H5(OH)3 in a solution of 36 g of water and 46 g of glycerine is :
Options
A.0.46
B.0.36
C.0.20
D.0.40
Solution
Mole of H2O = 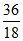 = 2
= 2
Mole of glycerine =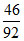 = 0.5
= 0.5
total mole = 2 + 0.5 = 2.5
Mole fractions of glycerine =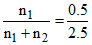
X0 = 0.2 Ans.
Mole of glycerine =
total mole = 2 + 0.5 = 2.5
Mole fractions of glycerine =
X0 = 0.2 Ans.
Create a free account to view solution
View Solution FreeMore Solution Questions
Each of the following pair shows a positive deviation from the Raoult’s law except...20 g of a substance were dissolved in 500 mL of water and the osmotic pressure of the solution was found to be 600 mm of...Mole fraction of a non-electrolyte in aqueous solution is 0.07. If Kf is 1.86o mol-1 kg, depression in f.p., ᐃTf, ...When during electrolysis of a solution of AgNO3 9650 coulombs of charge pass through the electroplating bath, the mass o...Which of the following is/are correct for an ideal binary solution of two volatile liquids (eg. benzene & toluene)?...