SolutionHard
Question
Under ambient conditions, which among the following surfactants will form micelles in aqueous solution at lowest molar concentration ?
Options
A.CH3−(CH2)8 −COO− Na+
B.CH3(CH2)11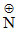 (CH3)3Br−
(CH3)3Br−
C.CH3−(CH2)13 − OSO3-Na+
D.CH3(CH2)15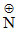 (CH3)3Br−
(CH3)3Br−
Solution
Longer hydrophobic chain, lesser CMC
Create a free account to view solution
View Solution FreeMore Solution Questions
The freezing point of equimolar aqueous solution will be highest for...When H2S gas is passed through the HCl containing aqueous solution of CuCl2, HgCl2, BiCl3, and CoCl2, Which does not pre...Consider the following four liquids :1. Water : highly polar ; H-bonding2. Hexanol : slightly polar ; some H-bonding3. C...Aluminium phosphate is 100% ionised in 0.01 molal aqueous solution. Hence, ᐃTb / Kb is :...Pure water boils at 373 K and nitric acid at 359 K. The azeotropic mixture of water and nitric acid boils at 393.5 K. On...