Gaseous StateHard
Question
If a gas is expanded at constant temperature :
Options
A.The pressure decrease
B.The kinetic energy of the molecules remains the same
C.The kinetic energy of the molecules decreases
D.the number of molecules of the gas increases
Solution
V ∞ 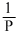 (Boyles, law)
(Boyles, law)
and K.E =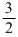 RT and it does not depend upon volume.
RT and it does not depend upon volume.
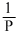 (Boyles, law)
(Boyles, law)and K.E =
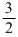 RT and it does not depend upon volume.
RT and it does not depend upon volume.Create a free account to view solution
View Solution FreeMore Gaseous State Questions
2.5 L of a sample of a gas at 27oC and 1 bar pressure is compressed to a volume of 500 mL keeping the temperature consta...The R.M.S. speed of the molecules of a gas of density 4 kg m-3 and pressure 1.2 ×105 N m- 2 is :...According to kinetic theory of gases, for a diatomic molecule :...The temperature at which a real gas obeys the ideal gas laws over a wide range of pressure is :...Compressibility factor for H2 behaving as real gas is :...