Gaseous StateHard
Question
According to kinetic theory of gases, for a diatomic molecule :
Options
A.the pressure exerted by the gas is proportional to mean valocity of the molecule
B.the pressure exerted by the gas is proportional to the root mean valocity of the molecule
C.the root mean square valocity of the molecule is inversely proportional to the temperature
D.the mean translational kinetic energy of the molecule is proprtonal to the absolute temperature
Solution
PV = 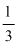 mnu2
mnu2
where u = root mean square velocity , m=mass of one molecule n= number of molecule
PV =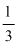 Mu2 (For one mole)
Mu2 (For one mole)
If n = NA, Then m × n = M (M. wt.)
and PV = RT (For one mole)
So,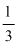 Mu2 = RT ⇒ Mu2 = 3RT
Mu2 = RT ⇒ Mu2 = 3RT
K. E. for one mole, Mu2 =
Mu2 = 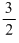 RT
RT
So K. E. / molecule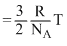
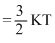
So K. E. ∝ T
 mnu2
mnu2where u = root mean square velocity , m=mass of one molecule n= number of molecule
PV =
 Mu2 (For one mole)
Mu2 (For one mole)If n = NA, Then m × n = M (M. wt.)
and PV = RT (For one mole)
So,
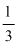 Mu2 = RT ⇒ Mu2 = 3RT
Mu2 = RT ⇒ Mu2 = 3RTK. E. for one mole,
 Mu2 =
Mu2 = 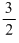 RT
RTSo K. E. / molecule
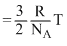
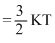
So K. E. ∝ T
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
Helium atom is two times heavier than a hydrogen molecule at 298 k, the average kinetic energy of helium is :...Which of the following statements are correct ?...One mole of a non-ideal gas undergoes a change of state (2.0 atm, 3.0L, 95K) → (4.0 atm, 5.0L, 245K) with a change...If a gas is allowed to expand at constant tempeature then which of the following does not hold true :...If two moles of an ideal gas at 546 K occupies a volume of 44.8 litres, the pressure must be -...