Gaseous StateHard
Question
Compressibility factor for H2 behaving as real gas is :
Options
A.1
B.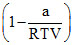
C.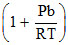
D.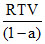
More Gaseous State Questions
For a real gas the P-V curve was experimentally plotted and it had the following appearance. With respect to liquifactio...By what factor does the average velocity of a gaseous molecule increase when the temperature (in Kelvin) is doubled ?...In vander Waal′s equation of state for a non ideal gas the term that accounts for intermolecular forces is :...One mole of a non-ideal gas undergoes a change of state (2.0 atm, 3.0L, 95K) → (4.0 atm, 5.0L, 245K) with a change...The transport of matter in the absence of bulk flow is known as :...