Gaseous StateHard
Question
In a solid AB having the NaCl structure, ′A′ atom occupies the corners of the cubic unit cell. If all the face-centred atoms along one axis are removed, then the resultant stoichiometry of the solid is :
Options
A.AB2
B.A2B
C.A4B3
D.A3B4
Solution
Na in NaCl has 8 corner and 6 face atoms. If we remove face centred atom of one axis, two face atoms are removed. Thus A is at 8 corner and B is at 4 faces.
∴ A =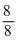 = 1 and B =
= 1 and B = 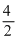 = 2
= 2
Thus formula is AB2
∴ A =
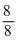 = 1 and B =
= 1 and B = 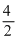 = 2
= 2Thus formula is AB2
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
According to the kinetic theory of gases, in an ideal gas, between two successive collisions a gas molecule travels...1 mol of an ideal gas at 27oC is compressed reverssibly from 20 L to 2L. Find out ᐃS :-...15 ml of a gaseous hydrocarbon was required for complete combustion in 357ml of air (21% of oxygen by volume) and the ga...When CO2 under high pressure is released from a fire extinguisher, particles of solid CO2 are formed, despite the low su...Van der waal′s equation (V - b) = nRT applicable for :...