Gaseous StateHard
Question
Van der waal′s equation 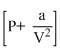 (V - b) = nRT applicable for :
(V - b) = nRT applicable for :
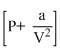 (V - b) = nRT applicable for :
(V - b) = nRT applicable for :Options
A.Ideal gas
B.Non-ideal gas
C.Both (a) and (b)
D.None of these
Solution
Vandar Waal′s equation is application for real (non-ideal)gasea.
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
NH3 gas is liquefied more easily than N2. Hence:...Consider given reaction : Metal (M) Select correct statement(s)...1g H2 gas at STP is expanded so that volume is doubled. Hence work done is :-...Two flasks of equal volume are connected by a narrow tube (of negligible volume) all at 27oC and contain 0.70 moles of H...Which of the following isothermal plots for fixed mass of ideal gas is / are correct ?...