Gaseous StateHard
Question
15 ml of a gaseous hydrocarbon was required for complete combustion in 357ml of air (21% of oxygen by volume) and the gaseous products occupied 327 ml (all volumes being measured at NTP). What is the formula of the hydrocarbon ?
Options
A.C3H8
B.C4H8
C.C5H10
D.C4H10
Solution
CxHy + O2 → xCO2 + 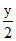 + H2O
+ H2O
15 ml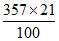 ml
ml
75 ml
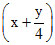 × 15 = 75 x +
× 15 = 75 x + 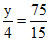
x +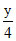 = 5 x +
= 5 x + 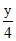 = 5
= 5
3 +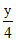 = 5 15 x + 15x + 282 = 327
= 5 15 x + 15x + 282 = 327
y = 8 x = 3
Formula = C3H8
15 ml
75 ml
x +
3 +
y = 8 x = 3
Formula = C3H8
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
The term that corrects for the attractive forces present in a real gas in the van der Waals equation is...The coordination number in hcp is...At STP the order of mean square velocity of molecules of H2, N2, O2 and HBr is -...Van der waal′s equation (V - b) = nRT applicable for :...Calculate the compressibility factor for CO2, if one mole of it occupies 0.4 litre at 300 K and 40 atm. Comment on the r...