Gaseous StateHard
Question
The rms velocity of hydrogen is √7 times the rms velocity of nitrogen. If T is the temperature of the gas :
Options
A.T(H2) = T(N2)
B.T(H2) > T(N2)
C.T(H2) < T(N2)
D.T(H2) = √7 T(N2)
Solution
urmsH2 = 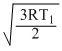
and urmsN2 =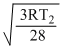
urmsH2 = √7 × urmsN2
or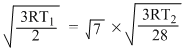
∴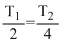
or T2 = 2T1 or TN2 > TH2
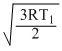
and urmsN2 =
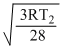
urmsH2 = √7 × urmsN2
or
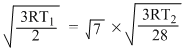
∴
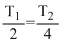
or T2 = 2T1 or TN2 > TH2
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
Given van der Waals constant for NH3, H2 and CO2 are respectively 4.17, 0.244, 1.36 and 3.59, which one of the following...For gaseous state, if most probable speed is denoted by C*, average speed by and mean square speed by C, then for a larg...A gaseous mixture containing He, CH4 & SO2 was allowed to effuse through a fine hole then find what molar ratio of gases...Henry′s law constants for four gases are given below at 293 K. Which one is more soluble in water at that temperat...N2 + 3H2 → 2NH3. 1 mol N2 and 4 mol H2 are taken in 15 L flask at 27oC. After complete conversion of N2 into NH3, ...