Gaseous StateHard
Question
N2 + 3H2 → 2NH3. 1 mol N2 and 4 mol H2 are taken in 15 L flask at 27oC. After complete conversion of N2 into NH3, 5 L of H2O is added. Pressure set up in the flask is :
Options
A.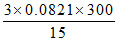 atm
atm
B.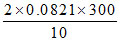 atm
atm
C.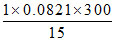 atm
atm
D.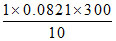 atm
atm
Solution
N2 + 3H2 → 2NH3
t = 0 1 mole 4 mole 0
t = tfinal 0 1 mole 2 mole
NH3 will absorb by water and volume will be 15 - 5 = 10 L
P =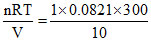 atm
atm
t = 0 1 mole 4 mole 0
t = tfinal 0 1 mole 2 mole
NH3 will absorb by water and volume will be 15 - 5 = 10 L
P =
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
For gaseous state, if most probable speed is denoted by C*, average speed by and mean square speed by C, then for a larg...0.28 g of a gas occupies 224 ml at STP. The gas could be...28. Gold has a face centered cubic lattice with an edge length of the unit cube of 407 pm. Assuming the closest packing ...A and B are two identical vessels. A contains 15 g ethane at 1atm and 298 K. The vessel B contains 75 g of a gas X2 at s...Three footballs are respectively filled with nitrogen, hydrogen and helium. If the leaking of the gas occurs with time f...