SolutionHard
Question
How many moles of sucrose should be dissolved in 500 gms of water so as to get a solution which has a difference of 104oC between boiling point and freezing point.
(Kf = 1.86 K Kg mol-1, Kb = 0.52 K Kg mol-1)
(Kf = 1.86 K Kg mol-1, Kb = 0.52 K Kg mol-1)
Options
A.1.68
B.3.36
C.8.40
D.0.840
Solution
Boiling point of solution = boiling point + ᐃTb = 100 + ᐃTb
Freezing point of solution = freezing point - ᐃTf = 0 - ᐃTf
Difference in temperature (given) = 100 + ᐃTb - (- ᐃTf)
104 = 100 + ᐃTb + ᐃTf = 100 + molality × Kb + molality × Kf
= 100 + molality (0.52 + 1.86)
∴ Molality =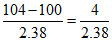 = 1.68 m
= 1.68 m
and molality =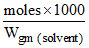 ; 1.68 =
; 1.68 = 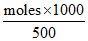
∴ Moles of solute =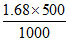 = 0.84 moles.
= 0.84 moles.
Freezing point of solution = freezing point - ᐃTf = 0 - ᐃTf
Difference in temperature (given) = 100 + ᐃTb - (- ᐃTf)
104 = 100 + ᐃTb + ᐃTf = 100 + molality × Kb + molality × Kf
= 100 + molality (0.52 + 1.86)
∴ Molality =
and molality =
∴ Moles of solute =
Create a free account to view solution
View Solution FreeMore Solution Questions
If P0 and PS, are the V.P. of solvent and its solution respectively and N1 and N2 are the mole fraction of solvent and s...The vapour pressure of a dilute aqueous solution of glucose is 750 mm of mercury at 373 K. The mole fraction of solute i...Consider the following aqueous solutions.I. 2.2 g Glucose in 125 mL of solution.II. 1.9 g Calcium chloride in 250 mL of ...A mixture contains 1 mole of volatile liquid A ($P_{A}^{o}$= 100 mm Hg) and 3 moles of volatile liquid B ($P_{B}^{o}$= 8...A quantity of 2 g of C6H5COOH dissolved in 25 g of benzene shows a depression in freezing point equal to 1.96 K. Molar d...