SolutionHard
Question
A solution of x moles of sucrose in 100 grams of water freezes at -0.2oC. As ice separates the freezing point goes down to 0.25oC. How many grams of ice would have separated?
Options
A.18 grams
B.20 grams
C.25 grams
D.23 grams
Solution
(i) ᐃTf = m × Kf
0.2 =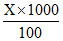 × 1.86 X =
× 1.86 X = 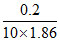
after freezing
ᐃTf = m × Kf
ᐃTf =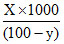 × 1.86 ᐃTf = 0.25
× 1.86 ᐃTf = 0.25
On solving, Amount of ice y = 20 g ice
0.2 =
after freezing
ᐃTf = m × Kf
ᐃTf =
On solving, Amount of ice y = 20 g ice
Create a free account to view solution
View Solution FreeMore Solution Questions
The boiling point and freezing point of a solvent ‘A’ are 90.0°C and 3.5°C, respectively. The Kf and Kb values of the so...Among the following, the surfactant that will form micelles in aqueous solution at the lowest molar concentration at amb...At particular temperature, the vapour pressure of two liquids A and B are respectively 120 and 180 mm of mercury. If 2 m...Gold number of haemoglobin is 0.03. Hence, 1000 mL of gold sol will require haemoglobinso that gold is not coagulated by...If P0 and P are the vapour pressures of a solvent and its solution respectively and N1 and N2 are the mole fractions of ...