SolutionHard
Question
What is the mole ratio of benzene (PB0 = 150 torr) and toluene (PT0 = 50 torr) in vapour phase if the given solution has a vapour pressure of 120 torr?
Options
A.7 : 1
B.7 : 3
C.8 : 1
D.7 : 8
Solution
P = PBoXB + PTo XT
120 = 150 (XB) + 50 (1 - XB)
100 XB = 70
XB = 0.7
YB =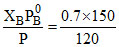 = 0.075
= 0.075 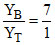 YT = 1 - 0.875 = 0.125
YT = 1 - 0.875 = 0.125
120 = 150 (XB) + 50 (1 - XB)
100 XB = 70
XB = 0.7
YB =
Create a free account to view solution
View Solution FreeMore Solution Questions
Select correct statement :...A complex containing K+, Pt(IV) and Cl- is 100% ionised giving i = 3. Thus, complex is :...The osmotic pressures of 0.010 M solutions of KI and of sucrose (C12H22O11) are 0.432 atm and 0.24 atm respectively. The...18 g of glucose (C6H12O6) is added to 178.2 g of water. The vapour pressure of water for this aqueous solution at 100oC ...Pure benzene freezes at 5.45o CA 0.374 m solution of terachloroethane in benzene freezes at 3.35o C. The Kf for benzene ...