SolutionHard
Question
18 g of glucose (C6H12O6) is added to 178.2 g of water. The vapour pressure of water for this aqueous solution at 100oC is
Options
A.759.00 Torr
B.7.60 Torr
C.76.00 Torr
D.752.40 Torr
Solution
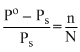
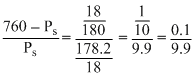
760 - Ps =
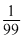 Ps
Ps 760 × 99 - Ps × 99 = Ps
760 × 99 = 100 Ps
Ps =
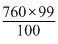 = 452.4
= 452.4Create a free account to view solution
View Solution FreeMore Solution Questions
Fog is a colloidal solution of:...Mole fraction of C3H5(OH)3 in a solution of 36 g of water and 46 g of glycerine is :...Which one of the following statements is false?...Which of the following curves represents the Henry′s law ?...The Ksp of AgI is 1.5 × 10-16 on mixing equal volumes of the following solutions precipitation will occur only with...